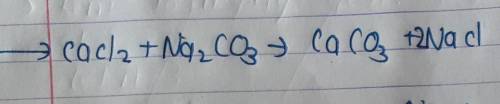Chemistry, 04.08.2020 14:01 shouyuanroh8462
Balance the following equation. Choose "blank" if no coefficient other than 1 is needed. CaCl2 + Na2CO3 CaCO3 + NaCl

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3

Chemistry, 22.06.2019 18:30
Read the claim. breakfast is an important meal. it jump starts the body’s process of using calories to break down food. appetite can decrease with age, but going too long without eating causes metabolism to slow down. current research shows that incorporating legumes such as lentils and chickpeas into meals boosts metabolism for twenty-four hours. who might benefit from this claim? people who have a fast metabolism stores that sell exercise equipment people who take vitamin supplements grocery stores that sell legumes
Answers: 1

You know the right answer?
Balance the following equation. Choose "blank" if no coefficient other than 1 is needed. CaCl2 + Na2...
Questions




Geography, 24.08.2019 17:30

Mathematics, 24.08.2019 17:30


Mathematics, 24.08.2019 17:30

Chemistry, 24.08.2019 17:30

Spanish, 24.08.2019 17:30






Mathematics, 24.08.2019 17:30

History, 24.08.2019 17:30

Mathematics, 24.08.2019 17:30