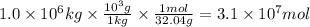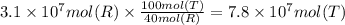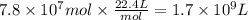Chemistry, 31.07.2020 08:01 daijahamaker062816
Methanol is produced industrially by catalytic hydrogenation of carbon monoxide according to the following equation: CO(g) + 2 H2(g) → CH3OH(l) If the yield of the reaction is 40%, what volume of CO (measured at STP) would be needed to produce 1.0 × 106 kg CH3OH?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:20
Why does his teacher ask him to balance the equation by including the correct coefficient
Answers: 1

Chemistry, 22.06.2019 19:00
Which statement best describes what happens when molecular compounds melt
Answers: 1

Chemistry, 22.06.2019 23:10
Using the periodic table, complete the following. element: hydrogen symbol: h₂ molecular weight: g mass of one mole: g/mol
Answers: 3

Chemistry, 23.06.2019 06:30
Acompound has the molecular formula c3h8. which class of organic compounds does it belong to?
Answers: 2
You know the right answer?
Methanol is produced industrially by catalytic hydrogenation of carbon monoxide according to the fol...
Questions



English, 30.05.2021 18:40

Mathematics, 30.05.2021 18:40


Computers and Technology, 30.05.2021 18:40


Mathematics, 30.05.2021 18:40

Computers and Technology, 30.05.2021 18:40


Mathematics, 30.05.2021 18:40

Mathematics, 30.05.2021 18:40

Social Studies, 30.05.2021 18:40


Mathematics, 30.05.2021 18:40


Social Studies, 30.05.2021 18:40

Chemistry, 30.05.2021 18:40

English, 30.05.2021 18:40

Mathematics, 30.05.2021 18:50