Chemistry, 30.07.2020 08:01 putaprincess16
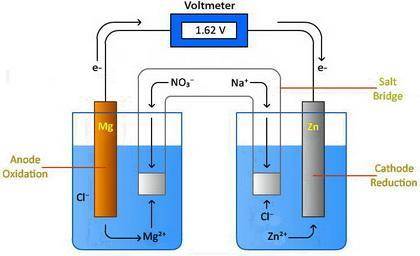
Design a voltaic cell using magnesium as one of the electrodes. Magnesium can be represented as either Metal A or Metal B in the above drawing. Use metal chlorides as the solutions in the two chambers. For example, magnesium chloride, (MgCl2) will be in solution in the chamber with the magnesium electrode. Use NaNO3 in the salt bridge. Select another element for the other electrode. Explain why you selected this element. Include information about the activity of the metal you select and the need for a spontaneous reaction. Metal A: Metal B: In the drawing, 1. Label the oxidation compartment: 2. Label the reduction compartment. 3. Label the direction of the flow of electrons. 4. Label the flow of the magnesium ions. 5. Label the flow of your selected element's ions. 6. What is leaving the salt bridge in the anode compartment? 7. What is leaving the salt bridge in the cathode compartment? 8. Write the oxidation and reduction half-reactions. 9. Calculate the chemical potential of your cell. Show all of your work.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Apeak with a retention time of 407 s has a width at half-height (w1/2) of 7.6 s. a neighboring peak is eluted 17 s later with a w1/2 of 9.4 s. a compound that is known not to be retained was eluted in 2.5 s. the peaks are not baseline resolved. how many theoretical plates would be needed to achieve a resolution of 1.5?
Answers: 2


Chemistry, 23.06.2019 04:50
The diagin dilutepage 6 of 12a6a5(a)fluorine, chlorine, bromine and iodine are placed in the same group of theperiodic table.state the common name used to describe elements in this group.(i)state the group in which the elements are placed and explain whythey are placed in that group.(ii)which of the above named elements is a solid at roomtemperature and pressure?
Answers: 2

You know the right answer?
Design a voltaic cell using magnesium as one of the electrodes. Magnesium can be represented as eith...
Questions

Mathematics, 04.07.2019 19:30

History, 04.07.2019 19:30

English, 04.07.2019 19:30

Mathematics, 04.07.2019 19:30


English, 04.07.2019 19:30


Chemistry, 04.07.2019 19:30

Mathematics, 04.07.2019 19:30

Mathematics, 04.07.2019 19:30

English, 04.07.2019 19:30



Mathematics, 04.07.2019 19:30

Mathematics, 04.07.2019 19:30

Biology, 04.07.2019 19:30

English, 04.07.2019 19:30


Mathematics, 04.07.2019 19:30

Mathematics, 04.07.2019 19:30