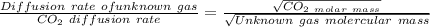Chemistry, 29.07.2020 21:01 Sparkleskeepsgoing
A gas of unknown identity diffuses at a rate of 155 mL/s in a diffusion apparatus in which carbon dioxide diffuses at the rate of 102 mL/s. Calculate the molecular mass of the unknown gas.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3


Chemistry, 22.06.2019 09:00
How are isotopes of the same chemical element alike? how are they different?
Answers: 1

Chemistry, 22.06.2019 10:50
A100 kmol/h stream that is 97 mole% carbon tetrachloride (ccl4) and 3% carbon disulfide (cs2) is to be recovered from the bottom of a distillation column. the feed to the column is 16 mole% cs2 and 84% ccl4, and 2% of the ccl4 entering the column is contained in the overhead stream leaving the top of the column. calculate the mass and mole fractions of ccl4 in the overhead stream, and determine the molar flow rates of ccl4 and cs2 in the overhead and feed streams. 12. mw_ccla- 153.82; mw_cs2-76.14.
Answers: 3
You know the right answer?
A gas of unknown identity diffuses at a rate of 155 mL/s in a diffusion apparatus in which carbon di...
Questions

History, 01.10.2019 14:10


Spanish, 01.10.2019 14:10


History, 01.10.2019 14:10


Mathematics, 01.10.2019 14:10

Advanced Placement (AP), 01.10.2019 14:10


Social Studies, 01.10.2019 14:10


Mathematics, 01.10.2019 14:10

Social Studies, 01.10.2019 14:10





Geography, 01.10.2019 14:20

Arts, 01.10.2019 14:20

Chemistry, 01.10.2019 14:20