
Chemistry, 29.07.2020 04:01 stacy021603
The Handbook of Chemistry and Physics gives solubilities of the following compounds in grams per 100 mL water. Because these compounds are only slightly soluble, assume that the volume does not change on dissolution and calculate the solubility product for each.
(a) BaSeO4, 0.0118 g/100 mL
(b) Ba(BrO3)2 H20, 0.30 g/100 mL
(c) NH4MgAsO4-6H20, 0.038 g/100 mL
(d) La2(MoOs)3, 0.00179 g/100 mL

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:30
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1

Chemistry, 22.06.2019 10:00
What is the atomic mass of an atom that has 6 protons, 6 neutrons, and 6 electrons? a) 6 b) 8 c) + 1 d) 12 e) 18
Answers: 1

Chemistry, 22.06.2019 13:30
Table sugar completely dissolved in water is an example of a?
Answers: 1

Chemistry, 22.06.2019 22:10
What is the indicator of the number of ions in solution? the amount of conductivity the amount of precipitate the amount of solute added
Answers: 1
You know the right answer?
The Handbook of Chemistry and Physics gives solubilities of the following compounds in grams per 100...
Questions

History, 24.11.2020 23:30


Mathematics, 24.11.2020 23:30


Computers and Technology, 24.11.2020 23:30

Mathematics, 24.11.2020 23:30

History, 24.11.2020 23:30


Mathematics, 24.11.2020 23:30


Spanish, 24.11.2020 23:30

Mathematics, 24.11.2020 23:30


Social Studies, 24.11.2020 23:30




Mathematics, 24.11.2020 23:30


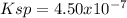
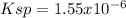
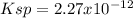
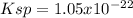
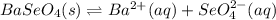
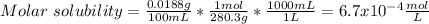
![Ksp=[Ba^{2+}][SeO_4^{2-}]=(6.7x10^{-4}\frac{mol}{L} )^2\\\\Ksp=4.50x10^{-7}](/tpl/images/0714/6084/c1a65.png)
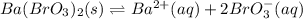
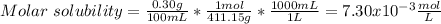
![Ksp=[Ba^{2+}][BrO_3^-]^2=(7.30x10^{-3}\frac{mol}{L})(3.65x10^{-3}\frac{mol}{L})^2\\\\Ksp=1.55x10^{-6}](/tpl/images/0714/6084/78dea.png)
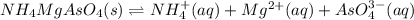
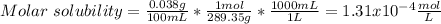
![Ksp=[NH_4^+][Mg^{2+}][AsO_4^{3-}]^2=(1.31x10^{-4}\frac{mol}{L})^3\\\\Ksp=2.27x10^{-12}](/tpl/images/0714/6084/beadb.png)
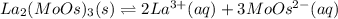
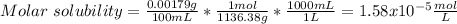
![Ksp=[La^{3+}]^2[MoOs^{-2}]^3=(2*1.58x10^{-5}\frac{mol}{L})^2(3*1.58x10^{-5}\frac{mol}{L})^3\\\\Ksp=1.05x10^{-22}](/tpl/images/0714/6084/a804d.png)


