Question 4
1 points
Save Answer
A solution is prepared at 25°C that is initially 0.42 M...

Chemistry, 23.07.2020 06:01 jaylen2559
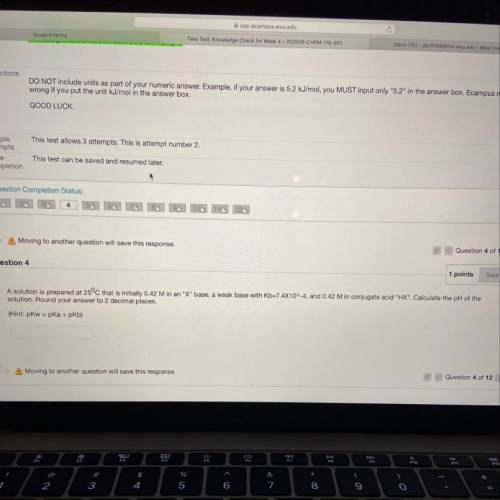
Question 4
1 points
Save Answer
A solution is prepared at 25°C that is initially 0.42 M in an "X" base, a weak base with Kb=7.4X10^-4, and 0.42 M in conjugate acid "HX". Calculate the pH of the
solution. Round your answer to 2 decimal places.
(Hint: pkw=pka + pkb)
A Moving to another question will save this response.
< Question 4 of 12>

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:30
What must happen before a body cell can begin mitotic cell division
Answers: 2


Chemistry, 23.06.2019 02:30
Ascientist wants to know how individual lions within a pride interact with each other in their own environment. to do this, the scientist sedates and tags all of the lions within a pride. then, he places several remotely-controlled video cameras near the lions' den and performs an observational field study. he collects continuous video footage over the span of one year, analyzes the video, and then forms conclusions based on his observations.
Answers: 2

Chemistry, 23.06.2019 11:30
How many grams of carbon are in 237 grams of ethanol(c2h5oh) and how many sulfide ions are in 2.45 moles of aluminum sulfide show me you you got the answers
Answers: 3
You know the right answer?
Questions

Mathematics, 21.03.2020 21:01



Mathematics, 21.03.2020 21:01

Mathematics, 21.03.2020 21:01








Chemistry, 21.03.2020 21:03









