Chemistry, 19.07.2020 17:01 Aurionna101
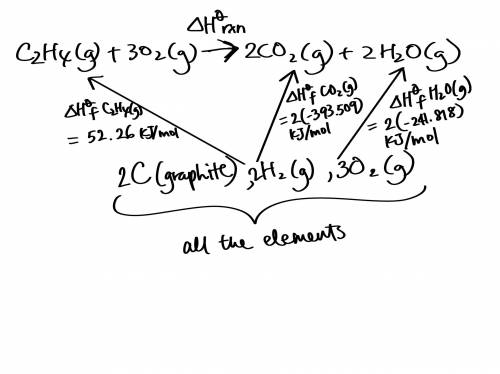
Use the standard enthalpies of formation for the reactants and products to solve for the ΔHrxn for the following reaction. (The ΔHf of C2H4 is 52.26 kJ/mol, CO2 is -393.509 kJ/mol, and H2O is -241.818 kJ.)
C2H4 (g) + 3O2(g) 2CO2 (g) + 2H2O(g)
ΔHrxn = (-345.64 kJ, -583.07 kJ, or -1,322.91 kJ).
The reaction is: (Endothermic or Exothermic).

Answers: 2


Another question on Chemistry


Chemistry, 21.06.2019 18:30
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible?
Answers: 2

Chemistry, 22.06.2019 10:10
When electrolyzing copper (ll) chloride, what reaction takes place at the anode? what reaction takes place at the cathode?
Answers: 1

You know the right answer?
Use the standard enthalpies of formation for the reactants and products to solve for the ΔHrxn for t...
Questions


Mathematics, 03.04.2020 17:58






Mathematics, 03.04.2020 17:59



Mathematics, 03.04.2020 17:59








English, 03.04.2020 18:00