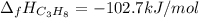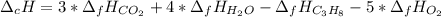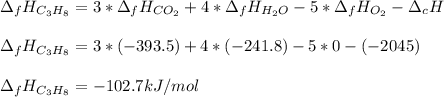Chemistry, 15.07.2020 01:01 jadahilbun01
The heat of reaction for the combustion of propane is –2,045 kJ. This reaction is: C3H8(g) +502 (g) 3CO2 (g) + 4H2O (g). Determine the standard heat of formation of propane.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:10
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3


Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 13:30
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2
You know the right answer?
The heat of reaction for the combustion of propane is –2,045 kJ. This reaction is: C3H8(g) +502 (g)...
Questions


Mathematics, 03.01.2022 01:10

Mathematics, 03.01.2022 01:20

Mathematics, 03.01.2022 01:20

Mathematics, 03.01.2022 01:20




Medicine, 03.01.2022 01:20


Mathematics, 03.01.2022 01:20

Social Studies, 03.01.2022 01:20

Mathematics, 03.01.2022 01:20


History, 03.01.2022 01:20



Social Studies, 03.01.2022 01:20

Advanced Placement (AP), 03.01.2022 01:20

Mathematics, 03.01.2022 01:20