
Chemistry, 13.07.2020 23:01 jeffhuffle17
The nature of zinc powder and Cobalt (II )oxide is heated the following reaction occurs ;
Zn(s)+CoO(s) => ZnO(s)+Co(s)
1)which metal is high in the reactivity series
2)the zink can be described as a reducing agent. using this Example explain what is the mean by the term ' reducing agent '
plz help!!!

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:40
In an effort to address concerns about global warming, a power plant in portland,oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

Chemistry, 22.06.2019 04:30
Why are people not able to scuba dive in the deep part of the ocean
Answers: 2

Chemistry, 22.06.2019 07:20
Which of these conditions most likely produces an unstable isotope?
Answers: 1

Chemistry, 22.06.2019 09:30
One way that radioactive waste is treated is by burying it in repositories. the repositories are found only in states with very low populations. true or false? a. trueb. false(also i meant to put high school but it put down middle school instead)
Answers: 1
You know the right answer?
The nature of zinc powder and Cobalt (II )oxide is heated the following reaction occurs ;
Zn(s)+CoO...
Questions

Mathematics, 30.01.2020 16:05

Geography, 30.01.2020 16:05

Chemistry, 30.01.2020 16:05







Mathematics, 30.01.2020 16:05

Mathematics, 30.01.2020 16:05


Mathematics, 30.01.2020 16:05

Biology, 30.01.2020 16:05

Mathematics, 30.01.2020 16:05


History, 30.01.2020 16:05


Mathematics, 30.01.2020 16:06

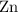 .
.

