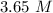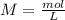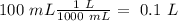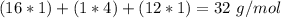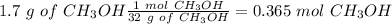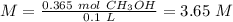Chemistry, 08.07.2020 19:01 ashley8057
What is the concentration (M) of CH3OH in a solution prepared by dissolving 11.7 g of CH3OH in sufficient water to give exactly 100. mL of solution

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10^-3 m and k for the dissociation is 1.86x10^-5. ch3cooh(aq)+h2o(> h3o^+(aq)+ch3coo^-(aq)
Answers: 2

Chemistry, 22.06.2019 06:10
56.16 gregor mendel was the first scientist to use statistics to analyze scientific data. before mendel's experiments, scientists believed that organisms acquired traits from their environment and passed them on to their offspring. after mendel's discoveries were accepted, scientists realized that traits passed to offspring were the result of genes being passed from parents to offspring. this is an example of pls
Answers: 1

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 12:00
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1
You know the right answer?
What is the concentration (M) of CH3OH in a solution prepared by dissolving 11.7 g of CH3OH in suffi...
Questions

Business, 13.10.2019 11:10

Mathematics, 13.10.2019 11:10

Mathematics, 13.10.2019 11:10

Health, 13.10.2019 11:10

Chemistry, 13.10.2019 11:10

Mathematics, 13.10.2019 11:10



Arts, 13.10.2019 11:10


Biology, 13.10.2019 11:10

English, 13.10.2019 11:10


Mathematics, 13.10.2019 11:10

History, 13.10.2019 11:10

Biology, 13.10.2019 11:10

Arts, 13.10.2019 11:10

Mathematics, 13.10.2019 11:10


Physics, 13.10.2019 11:10