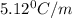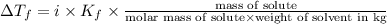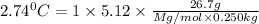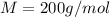Chemistry, 05.07.2020 14:01 queenjanet46
Suppose that you add 26.7 g of an unknown molecular compound to 0.250 kg of benzene, which has a K f of 5.12 oC/m. With the added solute, you find that there is a freezing point depression of 2.74 oC compared to pure benzene. What is the molar mass of the unknown compound

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:50
What are four significant sources of ghgs that come from wostem washington?
Answers: 2

Chemistry, 22.06.2019 10:00
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 11:00
The number to the right of an element's symbol (ex. c-12) identifies the of an isotope.
Answers: 1

You know the right answer?
Suppose that you add 26.7 g of an unknown molecular compound to 0.250 kg of benzene, which has a K f...
Questions


Physics, 13.07.2019 07:30

English, 13.07.2019 07:30



English, 13.07.2019 07:30

Biology, 13.07.2019 07:30




Mathematics, 13.07.2019 07:30




Social Studies, 13.07.2019 07:30

Mathematics, 13.07.2019 07:30


Spanish, 13.07.2019 07:30

Computers and Technology, 13.07.2019 07:30

Mathematics, 13.07.2019 07:30

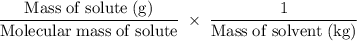
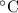
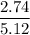 =
= 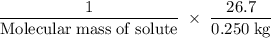
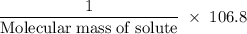
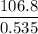 g/mol
g/mol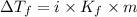
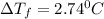 = Depression in freezing point
= Depression in freezing point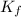 = freezing point constant =
= freezing point constant =