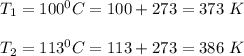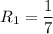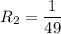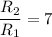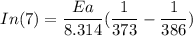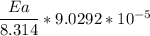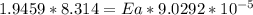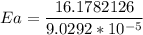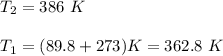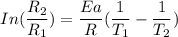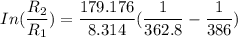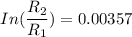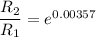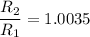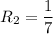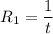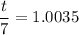Chemistry, 04.07.2020 14:01 landonp101
The water in a pressure cooker boils at a temperature greater than 100°C because it is under pressure. At this higher temperature, the chemical reactions associated with the cooking of food take place at a greater rate. (a) Some food cooks fully in 7.00 min in a pressure cooker at 113.0°C and in 49.0 minutes in an open pot at 100.0°C. Calculate the average activation energy for the reactions associated with the cooking of this food. kJ mol-1 (b) How long will the same food take to cook in an open pot of boiling water at an altitude of 10000 feet, where the boiling point of water is 89.8 °C? min

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:00
How does a hydroelectric power plant converts energy into energy.
Answers: 1

Chemistry, 23.06.2019 00:20
Which diagram represents the phase tha occurs after a solid melts?
Answers: 1

Chemistry, 23.06.2019 03:30
Ahelium balloon contains 16.9 l of helium at stp. how many atoms of helium are in the balloon
Answers: 1

Chemistry, 23.06.2019 13:00
Write the balanced chemical reaction for the formation of fe2(so4)3 from fe2o3 and so3 and determine how many moles of fe2(so4)3 are formed when 12.7 mol of so3 are reacted.
Answers: 1
You know the right answer?
The water in a pressure cooker boils at a temperature greater than 100°C because it is under pressur...
Questions



SAT, 07.10.2020 06:01


Mathematics, 07.10.2020 06:01



Mathematics, 07.10.2020 06:01



Health, 07.10.2020 06:01



Social Studies, 07.10.2020 06:01




Mathematics, 07.10.2020 06:01