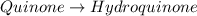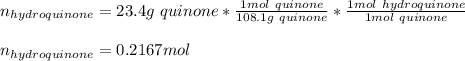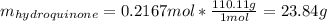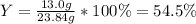Chemistry, 04.07.2020 01:01 allieb12334
Determine the theoretical maximum moles of hydroquinone, , that could be produced in this experiment. The reactant, quinone, is the limiting reagent. (To avoid introducing rounding errors on intermediate calculations, enter your answer to four significant figures.)
Reactant mass 23.4g
Product mass 13.0g
Reactant moles 0.2167 mol
Reactant mass 23.4g
Product mass 13.0g
Molar mass C 12.0 g/mol
Molar mass H 1.00 g/mol
Molar mass O 16.0 g/mol
Theoretical maximum moles of hydroquinone:

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Arock can be broken down into different kinds of substances by physical processes. no chemical reactions are needed to separate different parts of a rock into pure substances. this is because a rock is a(n)
Answers: 1


Chemistry, 22.06.2019 14:30
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3

Chemistry, 22.06.2019 17:30
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1
You know the right answer?
Determine the theoretical maximum moles of hydroquinone, , that could be produced in this experiment...
Questions





Computers and Technology, 25.11.2020 09:50

Mathematics, 25.11.2020 09:50

Mathematics, 25.11.2020 09:50

English, 25.11.2020 09:50

Health, 25.11.2020 09:50

Business, 25.11.2020 09:50


Mathematics, 25.11.2020 09:50



Mathematics, 25.11.2020 09:50

Chemistry, 25.11.2020 09:50



Computers and Technology, 25.11.2020 09:50