
Chemistry, 02.07.2020 17:01 emmaja121003
The value of ΔG°′ΔG°′ for the conversion of glucose-6-phosphate to fructose-6-phosphate (F6P) is +1.67 kJ/mol+1.67 kJ/mol . If the concentration of glucose-6-phosphate at equilibrium is 2.65 mM2.65 mM , what is the concentration of fructose-6-phosphate? Assume a temperature of 25.0°C25.0°C .

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:50
How will the emission of an alpha particle affect the atomic number of an atom
Answers: 3

Chemistry, 22.06.2019 08:30
Which part of earth’s surface receives the most direct rays from the sun? a) equator b) ocean c) poles d) mountains
Answers: 2

Chemistry, 22.06.2019 12:40
In the following table, all the columns for the element calcium are filled out correctly. element electron structure of atom electron structure of ion net ionic charge calcium 1s22s22p63s23p64s2 1s32s22p63s23p64s1 +1 true false
Answers: 2

Chemistry, 22.06.2019 15:00
Large helium-filled balloons are used to lift scientific equipment to high altitudes. what is the pressure inside such a balloon if it starts out at sea level with a temperature of 10.0ºc and rises to an altitude where its volume is twenty times the original volume and its temperature is – 50.0ºc ?
Answers: 2
You know the right answer?
The value of ΔG°′ΔG°′ for the conversion of glucose-6-phosphate to fructose-6-phosphate (F6P) is +1....
Questions

Biology, 01.09.2020 21:01



Social Studies, 01.09.2020 21:01

Mathematics, 01.09.2020 21:01

History, 01.09.2020 21:01

Biology, 01.09.2020 21:01

History, 01.09.2020 21:01

Mathematics, 01.09.2020 21:01

History, 01.09.2020 21:01

Mathematics, 01.09.2020 21:01

Chemistry, 01.09.2020 21:01

Mathematics, 01.09.2020 21:01

English, 01.09.2020 21:01

English, 01.09.2020 21:01

English, 01.09.2020 21:01

Mathematics, 01.09.2020 21:01


Mathematics, 01.09.2020 21:01

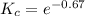
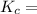 0.511
0.511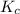
![K_c = \dfrac{[F6P]}{[G6P]}](/tpl/images/0699/9197/8414f.png)
![0.511 = \dfrac{[F6P]}{[2.65]}](/tpl/images/0699/9197/b9eb4.png)


