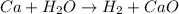Question 4
What is the sum of these two reactions? Write your answer below, but don't worry about formatting with subscripts-Do write chemical formulas with
proper atomic symbols, though (e. g., write CuCl2, not CUCL2).
(1) Ca + 2HCl - CaCl2 + H2
(2) CaCl2+H2O = CaO + 2HCl

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1

Chemistry, 22.06.2019 19:20
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3

Chemistry, 22.06.2019 20:10
Insoluble sulfide compounds are generally black in color. which of the following combinations could yield a black precipitate? check all that apply. na2s(aq)+kcl(aq) li2s(aq)+pb(no3)2(aq) pb(clo3)2(aq)+nano3(aq) agno3(aq)+kcl(aq) k2s(aq)+sn(no3)4(aq)
Answers: 1

Chemistry, 22.06.2019 21:20
One way in which the useful metal copper is produced is by dissolving the mineral azurite, which contains copper(ii) carbonate, in concentrated sulfuric acid. the sulfuric acid reacts with the copper(ii) carbonate to produce a blue solution of copper(ii) sulfate. scrap iron is then added to this solution, and pure copper metal precipitates out because of the following chemical reaction: (s) (aq) (s) (aq) suppose an industrial quality-control chemist analyzes a sample from a copper processing plant in the following way. he adds powdered iron to a copper(ii) sulfate sample from the plant until no more copper will precipitate. he then washes, dries, and weighs the precipitate, and finds that it has a mass of .
Answers: 2
You know the right answer?
Question 4
What is the sum of these two reactions? Write your answer below, but don't worry about...
Questions

History, 02.03.2020 00:09

Mathematics, 02.03.2020 00:09


Mathematics, 02.03.2020 00:09


Social Studies, 02.03.2020 00:09

Social Studies, 02.03.2020 00:09

Mathematics, 02.03.2020 00:10


English, 02.03.2020 00:10


English, 02.03.2020 00:10

Mathematics, 02.03.2020 00:10



History, 02.03.2020 00:10


Geography, 02.03.2020 00:11


Mathematics, 02.03.2020 00:11