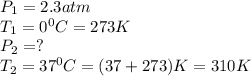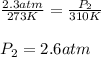2.A gas syringe at 20°C contains 20.0 mL of CO2 gas. The pressure of the gas in the syringe is 1.0 atm. What is the pressure in the syringe at 20°C if the plunger is depressed to 10.0 mL?
3.A sample of SO2 gas has a volume of 5.2 L. It is heated at constant pressure from 30.o oC to 90.0°C. What is its new volume? (Hint: Which law will you apply of the pressure is constant
4.A sample of the inhalation anesthetic gas Halothane, in a 500-mL cylinder has a pressure of 2.3 atm at 0°C. What will be the pressure of the gas if its temperature is warmed to 37°C (body temperature)? (Hint: Which law will you apply if the volume is constant?)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Why are people not able to skip a dive to the deepest part of the ocean
Answers: 1

Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1

Chemistry, 23.06.2019 00:00
#20 which type of bond is formed when bases pair in dna? ionic bond covalent bond coordinate bond hydrogen bond
Answers: 1

Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 1
You know the right answer?
2.A gas syringe at 20°C contains 20.0 mL of CO2 gas. The pressure of the gas in the syringe is 1.0 a...
Questions

History, 14.10.2019 13:00


Mathematics, 14.10.2019 13:00

Health, 14.10.2019 13:00


Biology, 14.10.2019 13:00


Mathematics, 14.10.2019 13:00

Chemistry, 14.10.2019 13:00

Mathematics, 14.10.2019 13:00

Mathematics, 14.10.2019 13:00

Business, 14.10.2019 13:00

Health, 14.10.2019 13:00


Mathematics, 14.10.2019 13:00

Biology, 14.10.2019 13:00



Mathematics, 14.10.2019 13:00

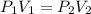
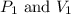 are initial pressure and volume.
are initial pressure and volume.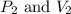 are final pressure and volume.
are final pressure and volume.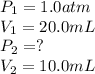
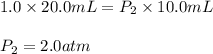
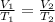
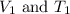 are the initial volume and temperature of the gas.
are the initial volume and temperature of the gas.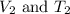 are the final volume and temperature of the gas.
are the final volume and temperature of the gas.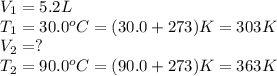
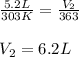
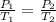
 are the initial pressure and temperature of the gas.
are the initial pressure and temperature of the gas. are the final pressure and temperature of the gas.
are the final pressure and temperature of the gas.