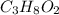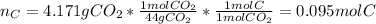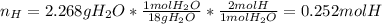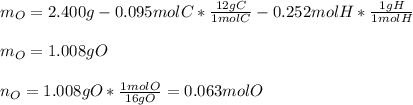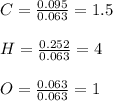Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the maximum amount of al2(so4)3 which could be formed from 15.84 g of al and 12.89 g of cuso4?
Answers: 2

Chemistry, 22.06.2019 09:00
Look at the spectrums of a star moving towards earth and a motionless star. which of these is a correct inference that can be draw from the observation of the two spectrums? (2 points) the spectrum of a motionless star is difficult to be viewed separately using oridinary telescopes. the spectrum of a motionless star is identical to the spectrum of a star which moves towards earth. the spectrum of a star shifts towards the red region when the star moves towards earth. the spectrum of a star shifts towards the blue region when the star moves towards earth.
Answers: 2

Chemistry, 22.06.2019 18:50
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1

You know the right answer?
Combustion analysis of 2.400 g of an unknown compound containing carbon, hydrogen, and oxygen produc...
Questions

Mathematics, 08.12.2020 23:50








Chemistry, 08.12.2020 23:50

Mathematics, 08.12.2020 23:50




Mathematics, 08.12.2020 23:50



Mathematics, 08.12.2020 23:50

English, 08.12.2020 23:50


Mathematics, 08.12.2020 23:50