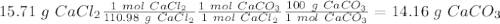Chemistry, 26.06.2020 15:01 smonahan2023
An excess of sodium carbonate, Na2CO3, in solution is added to a solution containing 15.71 g CaCl2. After performing the experiment, 13.19 g of calcium carbonate, CaCO3, is produced. Calculate the percent yield of this reaction

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Asyringe contains 56.05 ml of gas at 315.1 k. what volume will that gas occupy if the temperature is increased to 380.5 k? a) 12.41 b) 46.42 c) 67.68 d) 81.74
Answers: 1

Chemistry, 22.06.2019 03:30
What is the number of moles of chemical units represented by 9.03x10^24? and how do i show work? (dumb it down )
Answers: 1

Chemistry, 22.06.2019 07:20
Which of these conditions most likely produces an unstable isotope?
Answers: 1

Chemistry, 22.06.2019 08:00
Which of the following observations indicates that there is a small, dense, positively charged part in the center of an atom? some uncharged particles are scattered by a gold foil. all uncharged particles are attracted towards a gold foil. all positively charged particles pass straight through a gold foil. some positively charged particles bounce back from a gold foil.
Answers: 2
You know the right answer?
An excess of sodium carbonate, Na2CO3, in solution is added to a solution containing 15.71 g CaCl2....
Questions

History, 04.10.2019 19:00


Mathematics, 04.10.2019 19:00



Mathematics, 04.10.2019 19:00

Mathematics, 04.10.2019 19:00

Mathematics, 04.10.2019 19:00

Mathematics, 04.10.2019 19:00



Mathematics, 04.10.2019 19:00


Biology, 04.10.2019 19:00


History, 04.10.2019 19:00

Mathematics, 04.10.2019 19:00


English, 04.10.2019 19:00

Mathematics, 04.10.2019 19:00


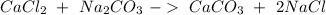
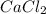 , so we have to do the following steps:
, so we have to do the following steps: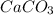 using the molar ratio. ( 1 mol
using the molar ratio. ( 1 mol