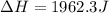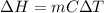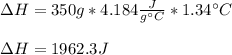Chemistry, 25.06.2020 09:01 christianmcafee
When 200g of AgNO3 solution mixes with 150 g of NaI solution, 2.93 g of AgI precipitates, and the temperature of the solution rises by 1.34oC. Assume 350 g of solution and a specific heat capacity of 4.184 J/g•oC. Calculate H for the following: Ag+(aq) + I- (aq) → AgI(s)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
How much heat is released upon converting one mole of steam (18.0 g) from 100.0 ∘c to water at 25.0 ∘c? show work and constants, trying to figure out how it works. only given the heat capacity for steam and water so try to only use that
Answers: 1


Chemistry, 22.06.2019 08:30
Analyze how limestone is weathered and identify the features that are formed as a result of this dissolution
Answers: 1

Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1
You know the right answer?
When 200g of AgNO3 solution mixes with 150 g of NaI solution, 2.93 g of AgI precipitates, and the te...
Questions



Mathematics, 04.08.2019 05:30



Physics, 04.08.2019 05:30



Mathematics, 04.08.2019 05:30


History, 04.08.2019 05:30

Social Studies, 04.08.2019 05:30




Mathematics, 04.08.2019 05:30



Chemistry, 04.08.2019 05:30