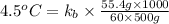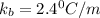Chemistry, 21.06.2020 06:57 ethanmatthews2336
A certain substance X condenses at a temperature of 120.7 degree C. But if a 500, g sample of X is prepared with 55.4 g of urea (NH_2)_2 CO) dissolved in it, the sample is found to have a condensation point of 125.2 degree C instead. Calculate the molal boiling point elevation constant K_b of X. Round your answer to 2 significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:30
When the earth was formed and cooled, why did nickel and iron end up in the center of the earth while basalt and granite ended up in the outer layers
Answers: 3

Chemistry, 22.06.2019 10:30
Find the number of grams of hcl needed to react completely with .50 moles of magnesium.
Answers: 1

Chemistry, 22.06.2019 15:00
Areaction is first order with respect to reactant x and second order with respect to reactant y. which statement describes the rate law for this reaction?
Answers: 1

Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 2
You know the right answer?
A certain substance X condenses at a temperature of 120.7 degree C. But if a 500, g sample of X is p...
Questions


History, 27.03.2020 19:15



Mathematics, 27.03.2020 19:15


History, 27.03.2020 19:15



Mathematics, 27.03.2020 19:16



Mathematics, 27.03.2020 19:16

Mathematics, 27.03.2020 19:16

Social Studies, 27.03.2020 19:16


Mathematics, 27.03.2020 19:16


History, 27.03.2020 19:16

Mathematics, 27.03.2020 19:16

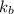 of X is
of X is 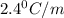
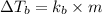
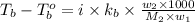
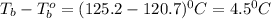
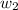 = mass of solute (urea) = 55.4 g
= mass of solute (urea) = 55.4 g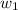 = mass of solvent X = 500 g
= mass of solvent X = 500 g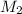 = molar mass of solute (urea) = 60 g/mol
= molar mass of solute (urea) = 60 g/mol