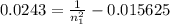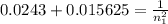Chemistry, 20.06.2020 18:57 tatianaware1617
The electron in a hydrogen atom, originally in level n = 8, undergoes a transition to a lower level by emitting a photon of wavelength 3745 nm. What is the final level of the electron? (c = 3.00 x 108m/s, h = 6.63 x 10−34 J • s, RH = 2.179 x 10−18 J)
a. 5
b. 6
c. 8
d. 9
e. 1

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 04:00
The continuous release of nuclear energy caused when one fission reaction triggered more nuclear reactions is a
Answers: 3

Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 14:10
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2
You know the right answer?
The electron in a hydrogen atom, originally in level n = 8, undergoes a transition to a lower level...
Questions


History, 06.07.2019 12:30




Mathematics, 06.07.2019 12:30


Mathematics, 06.07.2019 12:30

Mathematics, 06.07.2019 12:30



Mathematics, 06.07.2019 12:30



Mathematics, 06.07.2019 12:30


Geography, 06.07.2019 12:30

Mathematics, 06.07.2019 12:30

Mathematics, 06.07.2019 12:30

Mathematics, 06.07.2019 12:30

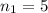
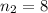
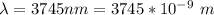
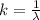
![k = R_{\infty} [\frac{1}{n_1^2} + \frac{1}{n_2^2} ]](/tpl/images/0690/9055/ac88e.png)
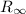 is the Rydberg constant, with a value
is the Rydberg constant, with a value 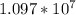
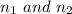 are the principal quantum levels
are the principal quantum levels ![0.0243= [\frac{1}{n_1^2} - \frac{1}{8^2} ]](/tpl/images/0690/9055/cbffd.png)