
Chemistry, 20.06.2020 08:57 graciewyatt471
1) How many kJ are absorbed when 45.2 g of water at 31.3 oC is heated to 76.9 oC? 2) Calculate the total heat released in kcal when 72.1 g water at 25.2 oC is cooled to 0 oC and freezes. 3) How many kilojoules are required to heat 55,500 mg of gold with specific heat = 0.129 J/g oC is heated from 24.6 oC to 123.4 oC? 4) Calculate the heat needed in kcal to change 45.6 g of water at 100 oC to change into steam.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:20
Give the orbital configuration of the phosphorus (p) atom.
Answers: 1

Chemistry, 22.06.2019 19:30
If 16.00g of hydrogen gas reacts with 126.73g of oxygen, how many grams of water are yielded? (both reactants are completely consumed in the reaction.)
Answers: 2


Chemistry, 23.06.2019 02:00
Pinene is an unsaturated hydrocarbon found in pine resin. if pinene has m+ = 136 and contains 1 double bond(s) and 2 ring(s); what is its molecular formula? enter the formula in the form ch first, then all other atoms in alphabetical order; do not use subscripts. the formula is case-sensitive
Answers: 3
You know the right answer?
1) How many kJ are absorbed when 45.2 g of water at 31.3 oC is heated to 76.9 oC? 2) Calculate the t...
Questions









Biology, 29.06.2020 20:01

Mathematics, 29.06.2020 20:01




Spanish, 29.06.2020 20:01


SAT, 29.06.2020 20:01

Mathematics, 29.06.2020 20:01



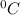 and Δθ is the change in temperature.
and Δθ is the change in temperature.

