Chemistry, 19.06.2020 03:57 extasisjorge
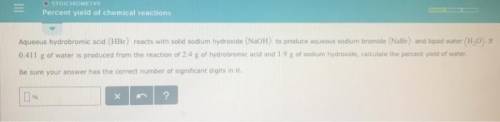
Aqueous hydrobromic acid reacts with solid sodium hydroxide to produce aqueous sodium bromide and liquid water . If of water is produced from the reaction of of hydrobromic acid and of sodium hydroxide, calculate the percent yield of water. Round your answer to significant figures.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Afamily is one another name for a group on the table of elements.
Answers: 1

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3


Chemistry, 23.06.2019 03:50
Show how to convert the temperature 84.7° c to kelvin. include all steps and label the final answer
Answers: 1
You know the right answer?
Aqueous hydrobromic acid reacts with solid sodium hydroxide to produce aqueous sodium bromide and li...
Questions

Health, 18.04.2021 15:30

English, 18.04.2021 15:30


Biology, 18.04.2021 15:30

Business, 18.04.2021 15:30

Chemistry, 18.04.2021 15:30


Computers and Technology, 18.04.2021 15:30

Physics, 18.04.2021 15:30



Social Studies, 18.04.2021 15:30

English, 18.04.2021 15:30



Physics, 18.04.2021 15:30

Mathematics, 18.04.2021 15:30

Mathematics, 18.04.2021 15:30