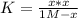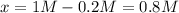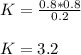Chemistry, 18.06.2020 05:57 andregijoe41
PCl5 dissociates according to the reaction: PCl5(g) = PCl3(g) + Cl2(g) One mole of PCl5 was placed in one liter of solution. When equilibrium was established, 0.2 mole of PCl5 remained in the mixture. What is the equilibrium constant for this reaction? (Hint: remember the ICE procedure? initial, change, and equilibrium)

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 18:30
You open a can of soda at room temperature and hear a hiss. which of the following factors has changed inside the container? a.) atmospheric pressure b.) temperature of gas c.) type of gas d.) amount of gas
Answers: 1

Chemistry, 23.06.2019 04:40
6) (a) calculate the absorbance of the solution if its concentration is 0.0278 m and its molar extinction coefficient is 35.9 l/(mol cm). the depth of the cell is 5 mm. (b) what is the %t? (7) calculate the absorbance of the solution if the transmitted light intensity is 70% of the initial light beam intensity
Answers: 1

Chemistry, 23.06.2019 08:30
What percentage of energy used in the u.s is produced from fossil fuels
Answers: 2
You know the right answer?
PCl5 dissociates according to the reaction: PCl5(g) = PCl3(g) + Cl2(g) One mole of PCl5 was placed i...
Questions


Arts, 24.02.2021 04:30

Mathematics, 24.02.2021 04:30


Mathematics, 24.02.2021 04:30



English, 24.02.2021 04:30





Mathematics, 24.02.2021 04:30


Computers and Technology, 24.02.2021 04:30




Mathematics, 24.02.2021 04:30

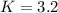
![K=\frac{[PCl_3][Cl_2]}{[PCl_5]}](/tpl/images/0688/6864/bc353.png)
 due to the reaction extent (ICE procedure):
due to the reaction extent (ICE procedure):