
8.50g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have a molecular molar mass of 176./gmol, is burned completely in excess oxygen, and the mass of the products carefully measured:
Product mass
Carbon dioxide 12.75g
Water 3.48g
Use this information to find the molecular formula of X.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:30
What happens to the atomic radius when an elctron is lost
Answers: 1

Chemistry, 22.06.2019 05:30
A3.37-mg sample of protein was chemically digested to convert its nitrogen into ammonia and then diluted to 100.0 ml. then 10.0 ml of this solution was placed in a 50-ml volumetric flask and treated with 5 ml of phenol solution plus 2 ml of sodium hypochlorite solution. the sample was diluted to 50.0 ml, and the absorbance at 625 nm was measured in a 1.00-cm cuvette and found to be 0.486. for reference, a standard solution was prepared from 10.0 mg of nh4cl (molar mass = 53.49 grams/mole) dissolved in 1.00 l of water. then 10.0 ml of this standard was placed in a 50-ml volumetric flask, treated in the same manner as the unknown, and the absorbance found to be 0.323. finally, a reagent blank was prepared using distilled water in place of unknown, it was treated in the same manner as the unknown, and the absorbance found to be 0.076. calculate the weight percent of nitrogen in the protein.
Answers: 1

Chemistry, 22.06.2019 13:20
Can someone me with 3 and 4 plz. this is for masteries test.
Answers: 2

Chemistry, 22.06.2019 21:00
Rays from the sun are not considered matter true or false
Answers: 2
You know the right answer?
8.50g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have...
Questions

Mathematics, 05.01.2020 09:31

Mathematics, 05.01.2020 09:31

Social Studies, 05.01.2020 09:31

Biology, 05.01.2020 09:31

Mathematics, 05.01.2020 09:31



Mathematics, 05.01.2020 09:31




Geography, 05.01.2020 09:31

Mathematics, 05.01.2020 09:31

Mathematics, 05.01.2020 09:31

Chemistry, 05.01.2020 09:31



Mathematics, 05.01.2020 09:31

Mathematics, 05.01.2020 09:31

History, 05.01.2020 09:31

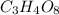
![[(CO_{2} )_{1.5} + (H_{2} O)_{1}]x](/tpl/images/0687/5815/1520d.png) = 176
= 176

