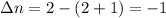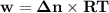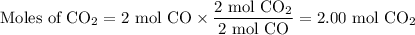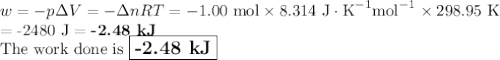Chemistry, 16.06.2020 02:57 estrellaalcantar16
Consider that you have a balloon containing 2.00 moles of CO and 1.00 mole of O2 which is in a room that has a temperature of 25.8oC and a pressure of 1.00 atm. Then, the following reaction occurs inside the balloon to completion: 2 CO(g) + O2(g) -> 2 CO2(g). Calculate the change in work due to the reaction occurring inside the balloon.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Several kinds of bears are found on earth. most bears are brown or black, but one type of bear, the polar bear, is white. what process led to this difference in fur color? explain your answer.
Answers: 1


Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 2

Chemistry, 22.06.2019 18:50
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1
You know the right answer?
Consider that you have a balloon containing 2.00 moles of CO and 1.00 mole of O2 which is in a room...
Questions



Physics, 06.03.2020 02:58




Mathematics, 06.03.2020 02:58

Computers and Technology, 06.03.2020 02:58





Mathematics, 06.03.2020 02:59


History, 06.03.2020 03:00





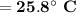
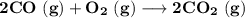
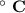 to
to 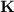 :
: 
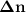 value:
value: