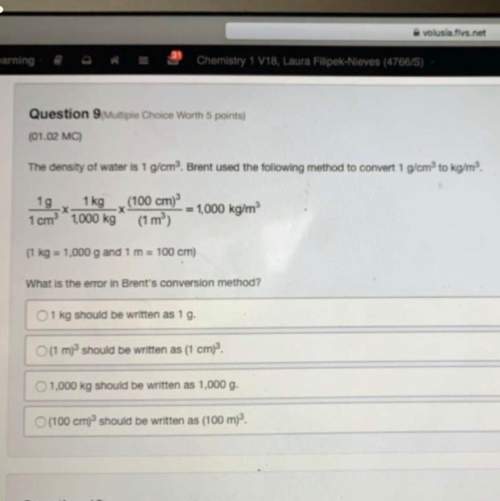
Chemistry, 13.06.2020 19:57 danielburke24
For the reaction NH4Cl (s)→NH3 (g) + HCl (g) at 25°C, ΔH = 176 kJ/moland ΔS = 0.285 kJ/(mol - K).
What is the ΔG, and will this reaction be spontaneous in the forward direction?
169 kJ/mol; no
91 kJ/mol; no
91 kJ/mol; yes
-91 kJ/mol; yes

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 20:30
What is a difference between a mixture of elements and a mixture of compounds
Answers: 1

Chemistry, 22.06.2019 21:20
The organs inside the body and how they function together
Answers: 3

Chemistry, 23.06.2019 00:00
What is the empirical formula of a compound that is 50.7% antimony and 49.3% selenium ?
Answers: 2
You know the right answer?
For the reaction NH4Cl (s)→NH3 (g) + HCl (g) at 25°C, ΔH = 176 kJ/moland ΔS = 0.285 kJ/(mol - K)....
Questions




Geography, 11.02.2020 22:44








Biology, 11.02.2020 22:45

Computers and Technology, 11.02.2020 22:45

Computers and Technology, 11.02.2020 22:45



Social Studies, 11.02.2020 22:45

English, 11.02.2020 22:45