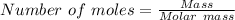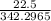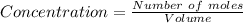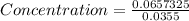Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 08:00
Which of the following observations indicates that there is a small, dense, positively charged part in the center of an atom? some uncharged particles are scattered by a gold foil. all uncharged particles are attracted towards a gold foil. all positively charged particles pass straight through a gold foil. some positively charged particles bounce back from a gold foil.
Answers: 2

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 12:10
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3
You know the right answer?
8) What is the molarity (M) of an aqueous solution containing 22.5 g of sucrose (C12H22011) in 35.5...
Questions



English, 09.10.2020 21:01



History, 09.10.2020 21:01

Computers and Technology, 09.10.2020 21:01

Chemistry, 09.10.2020 21:01



Social Studies, 09.10.2020 21:01


English, 09.10.2020 21:01


Mathematics, 09.10.2020 21:01

Mathematics, 09.10.2020 21:01



Computers and Technology, 09.10.2020 21:01

English, 09.10.2020 21:01