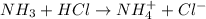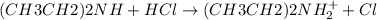Chemistry, 11.06.2020 19:57 memphissmith5779
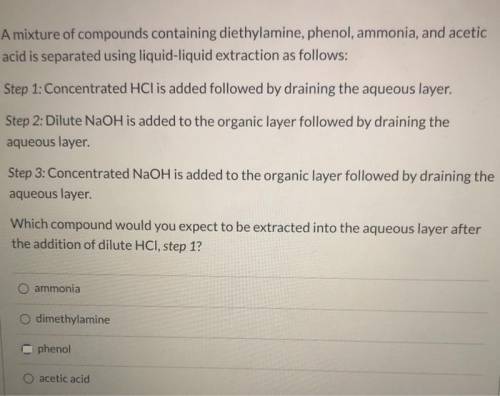
A mixture of compounds containing diethylamine, phenol, ammonia, and acetic acid is separated using liquid-liquid extraction as follows: Step 1: Concentrated HCl is added followed by draining the aqueous layer. Step 2: Dilute NaOH is added to the organic layer followed by draining the aqueous layer. Step 3: Concentrated NaOH is added to the organic layer followed by draining the aqueous layer. Which compound would you expect to be extracted into the aqueous layer after the addition of dilute HCl, step 1? Group of answer choices

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 19:30
How might this scientific phenomena be explained? a paper clip floats on water.
Answers: 1

Chemistry, 22.06.2019 23:30
If maltose undergoes hydrolysis what subunits does it results to?
Answers: 2

Chemistry, 23.06.2019 00:20
4. propanol and isopropanol are isomers. this means that they have a) the same molecular formula but different chemical properties. b) different molecular formulas but the same chemical properties. c) the same molecular formula and the same chemical properties. d) the same molecular formula but represent different states of the compound
Answers: 3

Chemistry, 23.06.2019 12:00
Which of the following statements is true? a. most heat energy is easily recovered and used for useful actions. b. friction causes molecules to vibrate more slowly. burning air and gasoline in an c. engine changes chemical energy into mechanical energy. it is impossible to d. change mechanical energy into mechanical energy.
Answers: 1
You know the right answer?
A mixture of compounds containing diethylamine, phenol, ammonia, and acetic acid is separated using...
Questions





History, 07.07.2019 21:20

Mathematics, 07.07.2019 21:20

Mathematics, 07.07.2019 21:20


Mathematics, 07.07.2019 21:20

Mathematics, 07.07.2019 21:20


Chemistry, 07.07.2019 21:20

Advanced Placement (AP), 07.07.2019 21:20

Mathematics, 07.07.2019 21:20


Biology, 07.07.2019 21:20




Biology, 07.07.2019 21:20