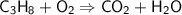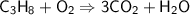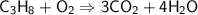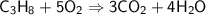Balance the combustion reaction between propane and oxygen.
C ₃ H ₈+ O ₂→ CO ₂ + H ₂ O...

Chemistry, 09.06.2020 20:57 avavackity
Balance the combustion reaction between propane and oxygen.
C ₃ H ₈+ O ₂→ CO ₂ + H ₂ O

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Arock can be broken down into different kinds of substances by physical processes. no chemical reactions are needed to separate different parts of a rock into pure substances. this is because a rock is a(n)
Answers: 1

Chemistry, 21.06.2019 18:00
How is the composition of a meteorite relevant to finding out the composition of earth's core?
Answers: 3

Chemistry, 21.06.2019 21:30
An alcohol thermometer makes use of alcohol's changing in order to measure temperature. as the temperature goes up, the alcohol contained in the thermometer increases in volume, filling more of the thermometer's tube.
Answers: 3

Chemistry, 22.06.2019 21:30
An atomic nucleus is composed ofa)protons.b)protons and neutrons.c)protons and electrons.d)protons, neutrons, and electrons.
Answers: 1
You know the right answer?
Questions


History, 20.09.2020 03:01


Mathematics, 20.09.2020 03:01

Mathematics, 20.09.2020 03:01


Mathematics, 20.09.2020 03:01



History, 20.09.2020 03:01

Mathematics, 20.09.2020 03:01

Social Studies, 20.09.2020 03:01






Chemistry, 20.09.2020 03:01


English, 20.09.2020 03:01


![\rule[225]{225}{2}](/tpl/images/0681/0254/85973.png)