
Chemistry, 10.06.2020 18:57 gretchcampbell
Acetic acid and ethanol react to form ethyl acetate and water. If 94.0 mmol of C2H5CO2CH3 are removed from a flask containing a mixture of HCH3CO2, C2H5OH, C2H5CO2CH3 and H2O at equilibrium, then following questions are to be answered. 1. What is the rate of the reverse reaction before any C2H5CO2CH3 has been removed from the flask?2. What is the rate of the reverse reaction just after the C2H5CO2CH3 has been removed from the flask?3. What is the rate of the reverse reaction when the system has again achieved equilibrium?4. How much less C2H5CO2CH3 is in the flask when the system has again reached equilibrium?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 19:30
What is the common name for the compound shown here? enter the common name of the compound shown?
Answers: 2

Chemistry, 22.06.2019 19:30
Astudent conducts an experiment to determine how the amount of water given to a plant affects its growth. what is the independent variable for this experiment?
Answers: 1

Chemistry, 22.06.2019 23:10
Match the formula for the following compound: magnesium sulfate heptahydratemgs·7h2omg2so4·7h2omg(so4)2·7h2omgso4·7h2o
Answers: 1
You know the right answer?
Acetic acid and ethanol react to form ethyl acetate and water. If 94.0 mmol of C2H5CO2CH3 are remove...
Questions

History, 03.03.2021 08:20

Geography, 03.03.2021 08:20




History, 03.03.2021 08:20

Mathematics, 03.03.2021 08:20






Chemistry, 03.03.2021 08:20






History, 03.03.2021 08:20

Mathematics, 03.03.2021 08:20

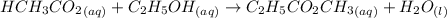
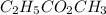 are removed from a flask; Then:
are removed from a flask; Then:

