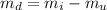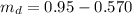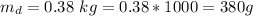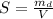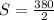Chemistry, 10.06.2020 06:57 cguzman4993
A chemistry student weighs out of an unknown solid compound and adds it to of distilled water at . After minutes of stirring, only some of the has dissolved. The student drains off the solution, then washes, dries and weighs the that did not dissolve. It weighs 0.570 kg.
Required:
a. Using the information above, can you calculate the solubility of X?
b. If so, calculate it. Remember to use the correct significant digits and units. .

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:00
Water is important for the of cells. a: size, shape, and temperature b: temperature, color, and odor c: color, odor, and size d: shape, temperature, and color
Answers: 2

Chemistry, 22.06.2019 04:30
Both josef loschmidt and amedeo avogadro contributed to our understanding of basic molecular numbers, sizes, and reaction ratios. neither scientist discovered “avogadro’s number” in the form we use it today (6.02 x 10 23). still, there’s a controversy over the name. research the contributions from these two scientists and read about how avogadro’s number got its name. briefly state what you think this number should be called, providing key details of each scientist’s contributions to this concept and a solid rationale for your case in naming the number.
Answers: 2

Chemistry, 22.06.2019 16:30
How many grams of mgbr2 are needed to produce 75g or metal?
Answers: 1

Chemistry, 23.06.2019 06:20
An object of mass 10.0 kg and volume 1000 ml and density 10 g/ml sinks in water who’s density is 1.0 g/ml. what is the mass of the water which has been displaced in kilograms
Answers: 1
You know the right answer?
A chemistry student weighs out of an unknown solid compound and adds it to of distilled water at . A...
Questions

Social Studies, 26.07.2019 12:30



Mathematics, 26.07.2019 12:30

Mathematics, 26.07.2019 12:30

English, 26.07.2019 12:30

Biology, 26.07.2019 12:40

Social Studies, 26.07.2019 12:40


Mathematics, 26.07.2019 12:40


History, 26.07.2019 12:40




History, 26.07.2019 12:40




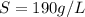
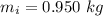
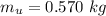
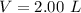
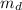 ) is mathematically represented as
) is mathematically represented as