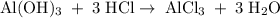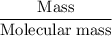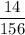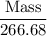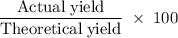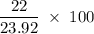Aluminum hydroxide is often present in antacids to neutralize stomach acid (HCl). If 14.0 g aluminum hydroxide is present in an antacid tablet, determine the theoretical yield of aluminum chloride produced when the tablet reacts with stomach acid. If the actual yield of the aluminum chloride from this tablet is 22.0 g, what is the percent yield?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 03:00
What happened in 2012 and how does it illustrate the importance of understanding the sun and how it works?
Answers: 3

Chemistry, 22.06.2019 03:30
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1

Chemistry, 22.06.2019 20:00
I’m an electrically neutral atomic any element, there are equal numbers of
Answers: 2
You know the right answer?
Aluminum hydroxide is often present in antacids to neutralize stomach acid (HCl). If 14.0 g aluminum...
Questions


Mathematics, 08.05.2021 01:00



Computers and Technology, 08.05.2021 01:00


Mathematics, 08.05.2021 01:00

Biology, 08.05.2021 01:00

Mathematics, 08.05.2021 01:00



Arts, 08.05.2021 01:00



Mathematics, 08.05.2021 01:00