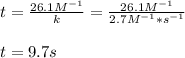Consider this reaction:
2Cl2O5 —> 2Cl2 + 5O2
At a certain temperature it obeys this...

Chemistry, 10.06.2020 04:57 michaellowe8434
Consider this reaction:
2Cl2O5 —> 2Cl2 + 5O2
At a certain temperature it obeys this rate law.
rate = (2.7.M^-1•s^-1) [Cl2O5]^2
Suppose a vessel contains Cl2O5 at a concentration of 0.600M. calculate how long it takes for the concentration of Cl2O5 to decrease by 94%. you may assume no other reaction is important. round your answer to two digits

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:10
Think about how you can use le chatelier’s principle to find possible solutions to the design problem. describe at least two ways to increase the yield (amount) of ammonia based on this principle.
Answers: 2

Chemistry, 22.06.2019 03:30
Explain why pure hydrogen cyanide does not conduct electricity, but become a conductor when it is dissolved in water? (at room temp, pure hcn exists as a volatile liquid)
Answers: 1

Chemistry, 22.06.2019 09:40
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2

Chemistry, 22.06.2019 10:30
Find the number of grams of hcl needed to react completely with .50 moles of magnesium.
Answers: 1
You know the right answer?
Questions



Mathematics, 03.07.2020 02:01





History, 03.07.2020 02:01




Biology, 03.07.2020 02:01


History, 03.07.2020 02:01



History, 03.07.2020 02:01

Mathematics, 03.07.2020 02:01

Mathematics, 03.07.2020 02:01

History, 03.07.2020 02:01

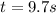
![\frac{1}{[Cl_2O_5]}=kt+\frac{1}{[Cl_2O_5]_0}](/tpl/images/0681/5190/837f8.png)
![[Cl_2O_5]=0.600M-0.600M*0.94=0.036M](/tpl/images/0681/5190/25d31.png)
![kt=\frac{1}{[Cl_2O_5]}-\frac{1}{[Cl_2O_5]_0}=\frac{1}{0.036M}-\frac{1}{0.60M} =26.1M^{-1}](/tpl/images/0681/5190/4b192.png)