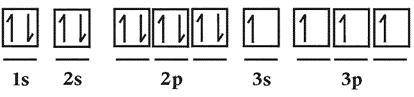Answers: 3


Another question on Chemistry



Chemistry, 23.06.2019 02:40
Calculate the standard enthalpy of formation of liquid methanol, ch3oh(l), using the following information: c(graphite) + o2 latex: \longrightarrow ⟶ co2(g) latex: \delta δ h° = –393.5 kj/mol h2(g) + o2 latex: \longrightarrow ⟶ h2o(l) latex: \delta δ h° = –285.8 kj/mol ch3oh(l) + o2(g) latex: \longrightarrow ⟶ co2(g) + 2h2o(l) latex: \delta δ h° = –726.4 kj/mol
Answers: 3

Chemistry, 23.06.2019 10:30
Me soon im confused much mass would a mole of hydrogen molecules contain? recall that hydrogen is diatomic. g/mol
Answers: 1
You know the right answer?
Please Help! I'm giving brainliest.
Which rrule for filling of oorbitals by electrons in the elemen...
Questions

English, 11.02.2021 03:10


History, 11.02.2021 03:10







Chemistry, 11.02.2021 03:10

Mathematics, 11.02.2021 03:10



Mathematics, 11.02.2021 03:10

History, 11.02.2021 03:10

Mathematics, 11.02.2021 03:10


Mathematics, 11.02.2021 03:10

Arts, 11.02.2021 03:10

Mathematics, 11.02.2021 03:10