Chemistry, 06.06.2020 20:58 kx85DirtbikeRider
The total enthalpy of the products in a reaction is 0 kJ, and the total enthalpy of the reactants is 100 kJ. What is the ΔH for the reaction? Choose the correct answer. –393.5 kJ –100 kJ 0 kJ +100 kJ

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Your friend offers to show you an intrusive igneous rock. which of the following would you expect to see?
Answers: 1

Chemistry, 22.06.2019 04:30
How much energy is made when a pice of wood burns. how do you know
Answers: 2

Chemistry, 23.06.2019 14:00
Tinererining 01: 57: 44 which statement correcte describes the actual veld and the theoretical yield of a reaction? textual vec is calculated to the reactant amounts but the theoretical yeld must be measured for each instance of a the actual vec is calculated from the amount of the limiting reactant and the theoretical yield is calculated from the 发公主 the actual weld depends on the reaction centers, but the theoretical yield and only with reactant amounts the actual vele represents the maximum weld possible and the theoretical yield assumes perfect reaction conditions save and ext e அட
Answers: 2

Chemistry, 23.06.2019 15:00
How many more valence electrons does sodium need to have a full outer valence shell
Answers: 3
You know the right answer?
The total enthalpy of the products in a reaction is 0 kJ, and the total enthalpy of the reactants is...
Questions

Mathematics, 07.12.2020 05:20


Mathematics, 07.12.2020 05:20

SAT, 07.12.2020 05:20


Mathematics, 07.12.2020 05:20

Biology, 07.12.2020 05:20

Mathematics, 07.12.2020 05:20

English, 07.12.2020 05:20



Mathematics, 07.12.2020 05:20

Mathematics, 07.12.2020 05:20

English, 07.12.2020 05:20



Mathematics, 07.12.2020 05:20

Chemistry, 07.12.2020 05:20


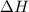 .
.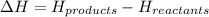
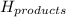 = enthalpy of products = 0 kJ
= enthalpy of products = 0 kJ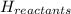 = enthalpy of reactants = 100 kJ
= enthalpy of reactants = 100 kJ