CaCO3(s)

Chemistry, 06.06.2020 08:58 CjTheStudentLOL
17. Consider the reaction shown and identify the statement that is not true.
825°C
CaCO3(s)
+ CaO(s) + CO2(g)
a.
This reaction is balanced as written.
b. The reactant must be heated for this reaction to occur.
c. The products are a solid and a gas.
d. Water must be present for this reaction to occur.
There are no solutions used in this reaction.
e.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 05:40
Consider the elements bromine and chlorine; which elements has a larger ionic radius ?
Answers: 1


Chemistry, 22.06.2019 16:50
Answer asap need it by wednesday morning calculate the ph of 0.02m hcl best answer will be brainliest
Answers: 1
You know the right answer?
17. Consider the reaction shown and identify the statement that is not true.
825°C
CaCO3(s)
CaCO3(s)
Questions



Chemistry, 25.02.2021 17:20




Social Studies, 25.02.2021 17:20

Mathematics, 25.02.2021 17:20

Mathematics, 25.02.2021 17:20

Mathematics, 25.02.2021 17:20


Mathematics, 25.02.2021 17:20

Mathematics, 25.02.2021 17:20

Spanish, 25.02.2021 17:20


Mathematics, 25.02.2021 17:20

Mathematics, 25.02.2021 17:20


Physics, 25.02.2021 17:20

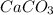 decomposition is:
decomposition is: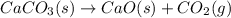
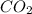 as product.
as product.

