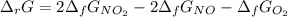(A) - 9.3 kcal

Chemistry, 06.06.2020 02:57 glowbaby123
Calculate the free energy change for the reaction:
2 NO(g) + O2(g) → 2 NO2(g)
(A) - 9.3 kcal
(B) + 24.9 kcal
(C) + 9.3 kcal
(D) - 16.6 kcal
(E) + 16.6 kcal


Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 13:30
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1

Chemistry, 22.06.2019 20:00
The picture represents the process that produces most of the energy used by living organisms on earth. which process is represented in the picture? a) the magnetic attraction between two hydrogen nuclei. b) the fusion of hydrogen nuclei to produce a helium nucleus in the core of the sun. c) the fission of hydrogen nuclei to produce a helium nucleus in the core of the sun. d) the chemical reaction between hydrogen nuclei to produce a helium nucleus in earth's atmosphere.
Answers: 3

Chemistry, 23.06.2019 07:00
Select the correct answer. why are scientific models important in the study of science? a. they always involve critical mathematical calculations. b. they scientists understand complex ideas and objects that aren’t easy to handle. c. they enable scientists to popularize their work in society. d. they are required when conducting any peer review process. e. they are necessary for turning a hypothesis into a law.
Answers: 2
You know the right answer?
Calculate the free energy change for the reaction:
2 NO(g) + O2(g) → 2 NO2(g)
(A) - 9.3 kcal
(A) - 9.3 kcal
Questions

History, 30.10.2020 17:30




English, 30.10.2020 17:30



Chemistry, 30.10.2020 17:30

English, 30.10.2020 17:30

Spanish, 30.10.2020 17:30





History, 30.10.2020 17:30





Computers and Technology, 30.10.2020 17:30