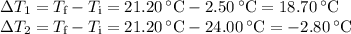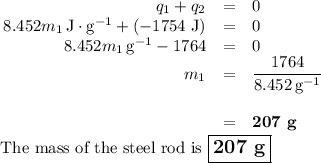CHEM EXPERT NEEDED***
A volume of 150.0 mL of H2O is initially at 24.00 °C. A chilled steel rod at 2.50 °C is placed in the water and the final temperature of the system is 21.20 °C.
Specific heat of water = 4.184 J/(g⋅∘C) and the specific heat of steel = 0.452 J/(g⋅∘C)
Write the equation and calculate the mass of the rod.

Answers: 2


Another question on Chemistry


Chemistry, 21.06.2019 19:00
Asmall amount of a solid is added to water. the observation made after fifteen minutes is shown in the figure. which of these solids has been probably added to water? a) oil b) sand c) sugar d) wood chips
Answers: 1


Chemistry, 22.06.2019 05:30
Choose all the answers that apply. as ocean depth increases, temperature decreases temperature increases pressure increases pressure decreases salinity increases density increases
Answers: 2
You know the right answer?
CHEM EXPERT NEEDED***
A volume of 150.0 mL of H2O is initially at 24.00 °C. A chilled steel rod at...
Questions

Mathematics, 18.07.2019 20:50

Biology, 18.07.2019 20:50


Biology, 18.07.2019 20:50









Spanish, 18.07.2019 20:50

Mathematics, 18.07.2019 20:50


Spanish, 18.07.2019 20:50




Biology, 18.07.2019 20:50