Chemistry, 04.06.2020 20:59 maysahdabest
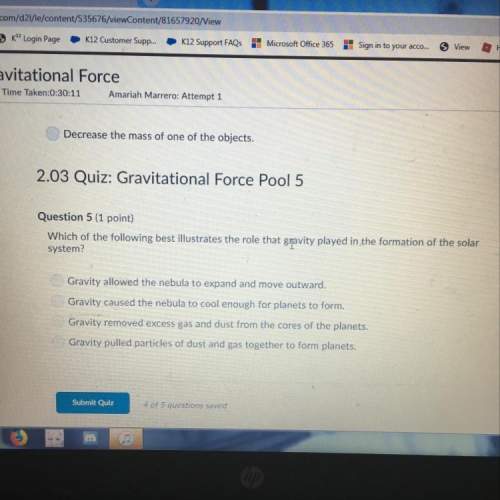
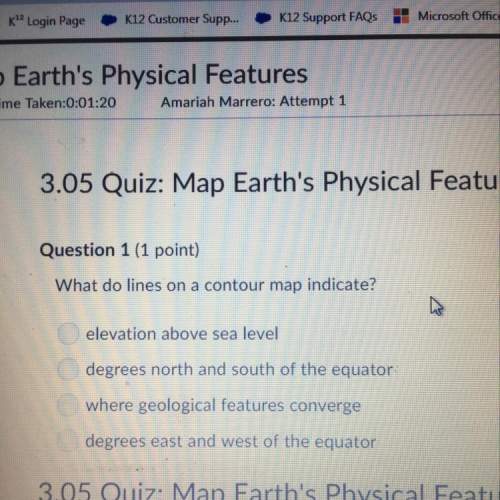
How many moles of a gas sample are in a 20.0 L container at 373 K and 203 kPa? The gas constant is 8.31 L−kPa/mol−K. A)0.33 mole B)0.65 mole C)1.30 moles D)3.05 moles

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:00
Drug abuse will not lead to physical and psychological dependence. true or false ?
Answers: 2


Chemistry, 23.06.2019 07:10
Which one of the following is an oxidation-reduction reaction? naoh + hno3 --> h2o + kno3 naoh + hno3 --> h2o + kno3 so3 + h2o --> h2so4 cacl2 + na2co3 --> caco3 + 2 nacl ch4 + 2 o2 --> co2 + 2 h2o al2(so4)3 + 6 koh --> 2 al(oh)3 + 3 k2so4
Answers: 3

Chemistry, 23.06.2019 14:00
If the molar mass of the compound is 96.69 g/mol, what is the molecular formula of the compound?
Answers: 1
You know the right answer?
How many moles of a gas sample are in a 20.0 L container at 373 K and 203 kPa? The gas constant is 8...
Questions





Computers and Technology, 30.11.2019 04:31





Computers and Technology, 30.11.2019 04:31