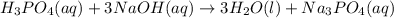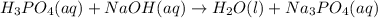Chemistry, 02.06.2020 23:00 angelinamadriga
In a neutralization reaction with a strong acid or base, all of the hydrogens from the acid and the hydroxides from the base react to form water.
Balance the following neutralization reaction:
H3PO4 (aq) + NaOH (aq) â H2O (l) + Na3PO4 (aq)
Enter the appropriate coefficient to the left of the respective term. In the event that your coefficient would be "1" be sure to write "1" as opposed to leaving it blank.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:00
A5.000 g sample of niso4 h2o decomposed to give 2.755 g of anhydrous niso4. what is the formula of the hydrate? what is the full chemical name for the hydrate? what is the molar mass of the hydrate? niso4•_h2o what is the mass % of water in the hydrate?
Answers: 1

Chemistry, 22.06.2019 14:30
The valence of aluminum is +3, and the valence of the chlorine is -1. the formula fir the aluminum chloride is correctly written as
Answers: 2


You know the right answer?
In a neutralization reaction with a strong acid or base, all of the hydrogens from the acid and the...
Questions

English, 29.10.2020 08:50

Mathematics, 29.10.2020 08:50

Spanish, 29.10.2020 08:50

Mathematics, 29.10.2020 08:50

English, 29.10.2020 08:50

Computers and Technology, 29.10.2020 08:50


English, 29.10.2020 08:50

Mathematics, 29.10.2020 08:50

History, 29.10.2020 08:50

Mathematics, 29.10.2020 08:50



Business, 29.10.2020 08:50


Chemistry, 29.10.2020 08:50

Mathematics, 29.10.2020 08:50



Law, 29.10.2020 08:50