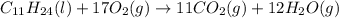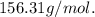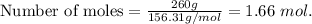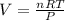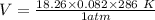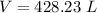Combustion of hydrocarbons such as undecane (C_11H_24) produces carbon dioxide, a "greenhouse gas." Greenhouse gases in the Earth's atmosphere can trap the Sun's heat, raising the average temperature of the Earth. For this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.
1. Write a balanced chemical equation, including physical state symbols, for the combustion of liquid undecane into gaseous carbon dioxide and gaseous water.
2. Suppose 0.260 kg of undecane are burned in air at a pressure of exactly 1 atm and a temperature of 13.0°C. Calculate the volume of carbon dioxide gas that is produced. Be sure your answer has the correct number of significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
What layer of the atmosphere is directly above the troposphere?
Answers: 1

Chemistry, 22.06.2019 09:00
The nuclear fission process releases neutrons and question 27 options: alpha particles electrons energy beta particles
Answers: 1

Chemistry, 22.06.2019 12:30
Which element has the lowest electronegativity? calcium(ca) gallium(ga) selenium(se) bromine(br)
Answers: 1

Chemistry, 22.06.2019 12:40
In the following table, all the columns for the element calcium are filled out correctly. element electron structure of atom electron structure of ion net ionic charge calcium 1s22s22p63s23p64s2 1s32s22p63s23p64s1 +1 true false
Answers: 2
You know the right answer?
Combustion of hydrocarbons such as undecane (C_11H_24) produces carbon dioxide, a "greenhouse gas."...
Questions

Biology, 05.10.2020 14:01

Mathematics, 05.10.2020 14:01


English, 05.10.2020 14:01


Mathematics, 05.10.2020 14:01

Mathematics, 05.10.2020 14:01


Mathematics, 05.10.2020 14:01



Biology, 05.10.2020 14:01

Mathematics, 05.10.2020 14:01


Business, 05.10.2020 14:01


Mathematics, 05.10.2020 14:01

Mathematics, 05.10.2020 14:01