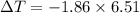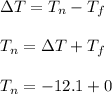What is the freezing point of a solution of 210.0 g of glycerol, formula C3H8O3, dissolved in 350. g of water? Careful. First get molar mass and use molar mass to determine molality concentration. Then use freeze pt. depression formula to determine the change in freezing pt. Then determine the new freeze point. The freezing point depression constant for water is Kf= -1.86 oCelcius/molal. Report your answer rounded to 1 decimal point and do not include units.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:00
A100-watt light bulb radiates energy at a rate of 100 j/s. (the watt, a unit of power or energy over time, is defined as 1 j/s.) if all of the light emitted has a wavelength of 525 nm , how many photons are emitted per second?
Answers: 1

Chemistry, 22.06.2019 09:30
Which formula can be used to calculate the molar mass of hydrogen peroxide
Answers: 1

Chemistry, 22.06.2019 12:00
What term is applied to a scientist who studies ancient life, including animal and plant fossils a. anthropologist b. dendroclimatologist c. geophysicist d. paleontologist
Answers: 2

You know the right answer?
What is the freezing point of a solution of 210.0 g of glycerol, formula C3H8O3, dissolved in 350. g...
Questions

Chemistry, 22.05.2020 08:04

Mathematics, 22.05.2020 08:04

Mathematics, 22.05.2020 08:04


Mathematics, 22.05.2020 08:04



Mathematics, 22.05.2020 08:04

History, 22.05.2020 08:04


Mathematics, 22.05.2020 08:04




Mathematics, 22.05.2020 08:04



Chemistry, 22.05.2020 08:04


Physics, 22.05.2020 08:04

 Freezing point depression constant for water, Kf = 0.512 °C/m
Freezing point depression constant for water, Kf = 0.512 °C/m

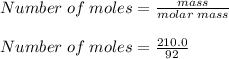
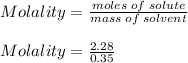
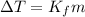
 is the change in temperature.
Kf is the molal freezing point constant.
m is the molality of solution.
is the change in temperature.
Kf is the molal freezing point constant.
m is the molality of solution.