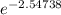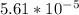Use one of your experimentally determined values of k, the activation energy you determined, and the Arrhenius equation to calculate the value of the rate constant at 25 °C. Alternatively, you can simply extrapolate the straight-line plot of ln(k) vs. 1/T in your notebook to 1/298, read off the value of ln(k), and determine the value of k. Please put your answer in scientific notation. slope=-12070, Ea=100kJ/mol, k= 0.000717 (45 °C), 0.00284 (55 °C), 0.00492 (65 °C), 0.0165 (75 °C), 0.0396 (85 °C).

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:40
Astudent made the lewis dot diagram of a compound shown. what is the error in the lewis dot diagram? a)an o atom should transfer all of its six electrons to mg because the formula is mgo b) both electrons of mg should be transferred to one o adam because the formula is mgo c) the electrons should be transferred from each o add him to capital mg because mg has fewer electrons d) the number of dots around mg should be four because it has to transfer two electrons to each o
Answers: 1

Chemistry, 22.06.2019 09:30
Apump contains 0.5 l of air at 203 kpa.you draw back on the piston of the pump, expanding the volume until the pressure reads 50.8 kpa. what is the new volume of the air pump
Answers: 2

Chemistry, 22.06.2019 13:30
What produces wave a)sound b) heats c)transfer of energy d)vibrations
Answers: 2

You know the right answer?
Use one of your experimentally determined values of k, the activation energy you determined, and the...
Questions


English, 04.11.2020 06:20



Mathematics, 04.11.2020 06:20

Mathematics, 04.11.2020 06:20


Mathematics, 04.11.2020 06:20

Mathematics, 04.11.2020 06:20

Mathematics, 04.11.2020 06:20

Mathematics, 04.11.2020 06:20



Business, 04.11.2020 06:20

Mathematics, 04.11.2020 06:20




English, 04.11.2020 06:20