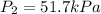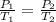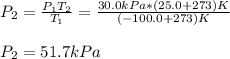Chemistry, 30.05.2020 19:02 hectorav6619
The temperature of a sample of gas in a steel
tank at 30.0 kPa is increased from -100.0°C to
25.0 °C. What is the final pressure inside the
tank?
A. 5.17 kPa
B. 51.7 kPa
C. 517 kPa
D. 5170 kPa

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:30
Aradio signal from a gps satellite take only about 0.067 seconds to reach a gps reciever. if the speed of light is about 300,000km/s, then approximately how far away is the reciever from from the satellite?
Answers: 1

Chemistry, 22.06.2019 17:20
Which of these features are formed when hot groundwater is forced out through cracks in the earth's surface?
Answers: 2

Chemistry, 23.06.2019 01:30
Magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. trial 1: trial 2: data mass of empty crucible with lid trial 1: 26.688 trial 2: 26.681 mass of mg metal, crucible, and lid trial 1: 26.994 trial: 2 26.985 mass of mgo, crucible, and lid trial 1: 27.188 trial 2: 27.180
Answers: 1

Chemistry, 23.06.2019 10:00
Which number should be placed before f2 on the reactants side equation to make equation balanced? xe + > xef4
Answers: 1
You know the right answer?
The temperature of a sample of gas in a steel
tank at 30.0 kPa is increased from -100.0°...
tank at 30.0 kPa is increased from -100.0°...
Questions

Social Studies, 02.01.2022 02:20

Mathematics, 02.01.2022 02:20

Mathematics, 02.01.2022 02:20


History, 02.01.2022 02:20

Advanced Placement (AP), 02.01.2022 02:20



History, 02.01.2022 02:20

Mathematics, 02.01.2022 02:20

Mathematics, 02.01.2022 02:20

World Languages, 02.01.2022 02:20


Computers and Technology, 02.01.2022 02:20

Physics, 02.01.2022 02:20


Mathematics, 02.01.2022 02:30