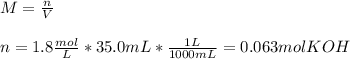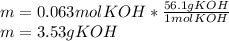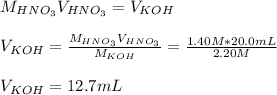Chemistry, 27.05.2020 23:01 sneakersolequeen
PLEASE SHOW WORK
1) You have a solution of acetic acid that has a K a of 3.5 × 10 –8 . If the concentration of the acetic acid were 2.40 M, what would be the concentration of H + at equilibrium?
2) You have a solution that is 1.75 M HCN. If the K a is 9.9 × 10 –8 , calculate the pH of the solution.
3) How many grams of KOH are needed to neutralize 35.0 mL of 1.8 M HCl?
4) How many mL of 2.20 M KOH are needed to neutralize 20.0 mL of 1.40 M HNO 3 ?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 05:30
Describe the interaction that occurs between two objects with the same electrical charge.
Answers: 1


Chemistry, 22.06.2019 09:40
How many grams of aluminum will there be in 98g of al2o3?
Answers: 1
You know the right answer?
PLEASE SHOW WORK
1) You have a solution of acetic acid that has a K a of 3.5 × 10 –8 . If the...
1) You have a solution of acetic acid that has a K a of 3.5 × 10 –8 . If the...
Questions



Geography, 25.07.2019 13:00

Mathematics, 25.07.2019 13:00

Mathematics, 25.07.2019 13:00



Health, 25.07.2019 13:00



Mathematics, 25.07.2019 13:00




History, 25.07.2019 13:00

Mathematics, 25.07.2019 13:00

Biology, 25.07.2019 13:00

Business, 25.07.2019 13:00

History, 25.07.2019 13:00

History, 25.07.2019 13:00

![[H^+]_{eq}=0.00029M](/tpl/images/0667/7635/6d441.png)
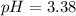
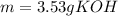
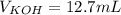
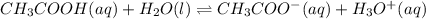
![Ka=\frac{[CH_3COO^-][H_3O^+]}{[CH_3COOH]}](/tpl/images/0667/7635/3cdd5.png)
 due to reaction's extent we have:
due to reaction's extent we have: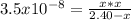
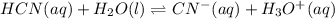
![K=\frac{[CN^-][H_3O^+]}{[HCN]}](/tpl/images/0667/7635/eab5d.png)
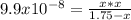
![[H^+]_{eq}=0.000416 M](/tpl/images/0667/7635/3b132.png)
![pH=-log([H^+]_{eq})=-log(0.000416)\\\\pH=3.38](/tpl/images/0667/7635/c9e0e.png)