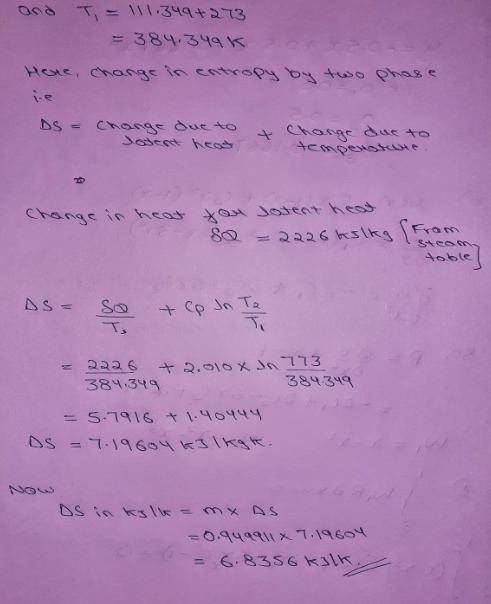Chemistry, 28.05.2020 19:01 donaldplawlerp5cctt
An insulated piston-cylinder device contains 1 L of saturated liquid water at pressure of 150 kPa. An electric resistance heater inside the cylinder is now turned on, and 2869.47 kJ of heat is transferred to the water. The inside H2O pressure maintains constant at 150 kPa during the process. Determine: the entropy change of the water during this heating process, in kJ/K.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:30
Label a-f based on the table using c for concentrated and d for dilute
Answers: 2

Chemistry, 22.06.2019 18:30
Which of the following nuclei would be the least stable a 2 protons, 2 neutrons b 1 proton 1 neutron c 1 proton 3 neutrons d 1 proton 2 neutrons
Answers: 3


You know the right answer?
An insulated piston-cylinder device contains 1 L of saturated liquid water at pressure of 150 kPa. A...
Questions

Engineering, 07.12.2021 01:00

History, 07.12.2021 01:00

Health, 07.12.2021 01:00





Mathematics, 07.12.2021 01:00



Mathematics, 07.12.2021 01:00

Mathematics, 07.12.2021 01:00

Computers and Technology, 07.12.2021 01:00

Medicine, 07.12.2021 01:00

Geography, 07.12.2021 01:00

Business, 07.12.2021 01:00

Mathematics, 07.12.2021 01:00