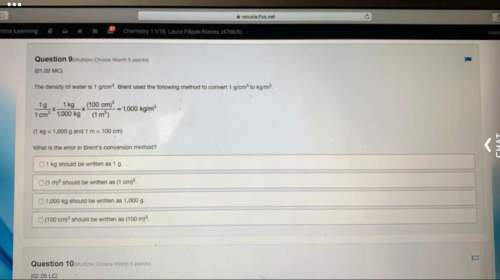
Chemistry, 28.05.2020 08:00 alexsysantiago54
K (potassium) belongs to group IA of the periodic table, and has 1 valence electron. Br (bromine) belongs to VIIA, and has
valence electrons.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:50
What is the composition, in atom percent, of an alloy that consists of 4.5 wt% pb and 95.5 wt% sn? the atomic weights for pb and sn are 207.19 g/mol and 118.71 g/mol, respectively.(a) 2.6 at% pb and 97.4 at% sn(b) 7.6 at% pb and 92.4 at% sn(c)97.4 at% pb and 2.6 at% sn(d) 92.4 at% pb and 7.6 at% sn
Answers: 2

Chemistry, 22.06.2019 10:10
What shape would a molecule with two bound groups and two lone pairs have?
Answers: 1

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 2

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2
You know the right answer?
K (potassium) belongs to group IA of the periodic table, and has 1 valence electron. Br (bromine) be...
Questions

History, 14.01.2021 01:40



Arts, 14.01.2021 01:40

History, 14.01.2021 01:40

Spanish, 14.01.2021 01:40


Advanced Placement (AP), 14.01.2021 01:40



Biology, 14.01.2021 01:40

Biology, 14.01.2021 01:40




Mathematics, 14.01.2021 01:50

English, 14.01.2021 01:50

Mathematics, 14.01.2021 01:50