Reaction A

Chemistry, 26.05.2020 14:57 jthollis1348
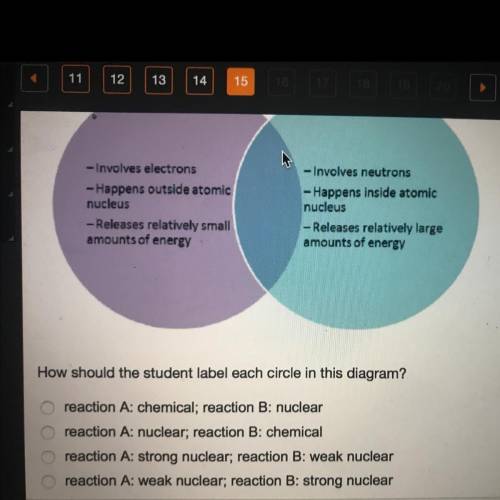
A student who is studying atomic reactions creates the following Venn diagram.
Reaction A
- Involves electrons
-Happens outside atomic
nucleus
- Releases relatively small
amounts of energy.
Reaction b - Involves neutrons
- Happens inside atomic
nucleus
- Releases relatively large
amounts of energy

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical solutions?
Answers: 1

Chemistry, 22.06.2019 18:00
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1
You know the right answer?
A student who is studying atomic reactions creates the following Venn diagram.
Reaction A
Reaction A
Questions

History, 01.04.2020 17:39



Mathematics, 01.04.2020 17:39

Mathematics, 01.04.2020 17:39




Social Studies, 01.04.2020 17:40




Mathematics, 01.04.2020 17:40

Mathematics, 01.04.2020 17:40


Mathematics, 01.04.2020 17:40

History, 01.04.2020 17:40

History, 01.04.2020 17:40




