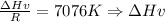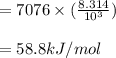6.4. The vapor pressure of ethylene glycol at several temperatures is given below:
T (C...

Chemistry, 22.05.2020 01:07 jones501324
6.4. The vapor pressure of ethylene glycol at several temperatures is given below:
T (C) 79.7 105.8 120.0 141.8 178.5 197.3
p* (mmHg) 5.0 20.0 40.0 100.0 400.0 760.0
Use a semilog plot based on the Clausius-Clapeyron equation to derive an equation for p* (mm Hg) as a function of T (C). From the plot, estimate the heat of vaporization of ethylene glycol in kJ/mol.
Clausius-Clapeyron equation: ln p* = -Hv/RT + B

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2


Chemistry, 23.06.2019 09:00
What factor besides temperature affects the boiling point of water? a. mass b. number of moles c. volume d. pressure
Answers: 3
You know the right answer?
Questions

Social Studies, 30.07.2019 07:00

English, 30.07.2019 07:00

Advanced Placement (AP), 30.07.2019 07:00

Social Studies, 30.07.2019 07:00


Business, 30.07.2019 07:00






Biology, 30.07.2019 07:00





Biology, 30.07.2019 07:00

Biology, 30.07.2019 07:00

Biology, 30.07.2019 07:00

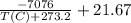
![Exp[\frac{-7076}{T(C)+273.2}+21.67 ]](/tpl/images/0660/5678/69715.png)